Hematological VitroScreen
- Preclinical Oncology CRO Services | Clinical Trial Specialty Testing
- Hematological VitroScreen
Industry-leading Hematologic Model Screening
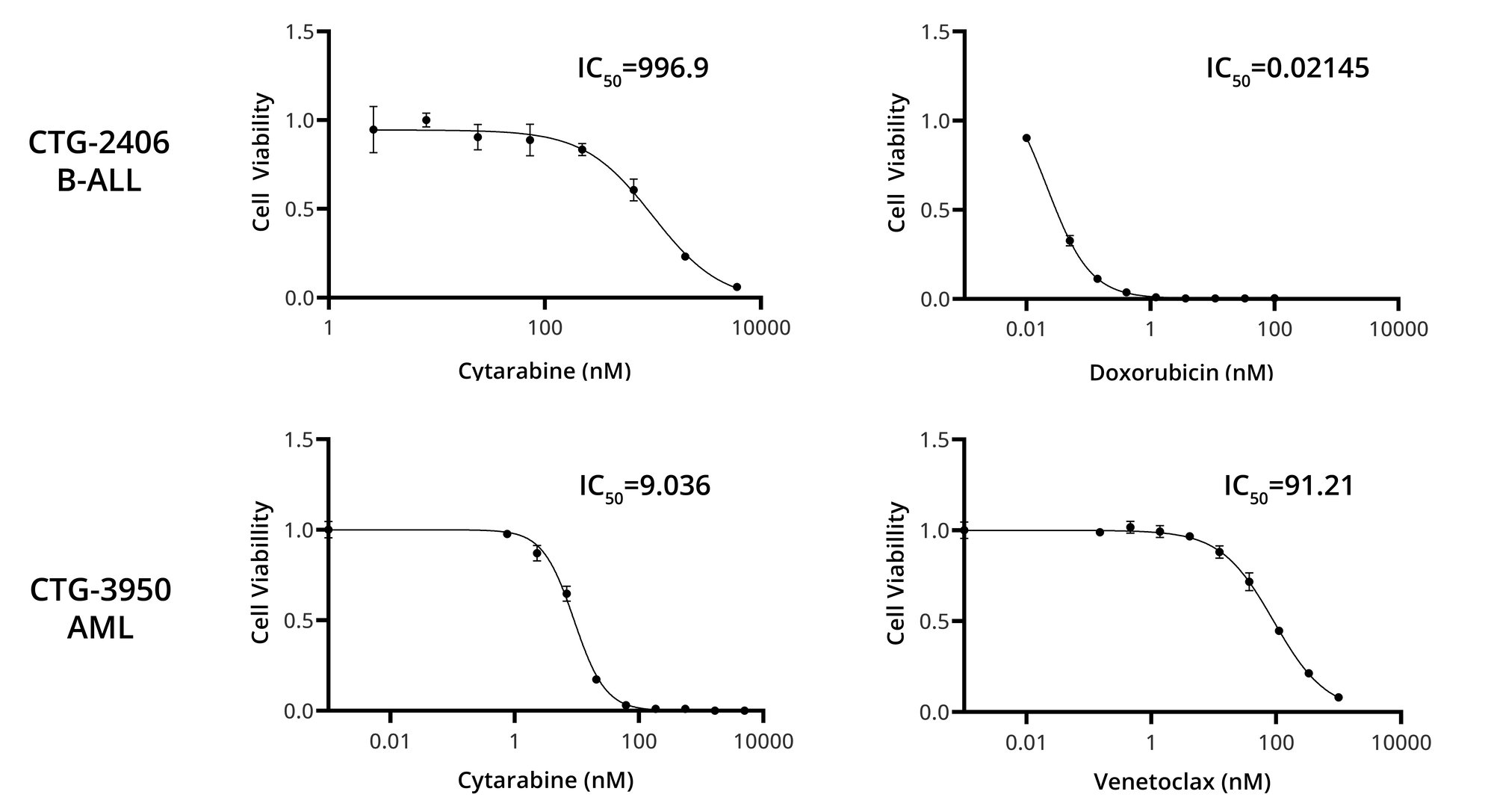
Champions' Hematological VitroScreens offer a quick, cost-effective solution to test your therapeutics in an ex vivo hematologic assay evaluating efficacy and mechanisms of action in primary patient samples.
-
Largest primary hematological repository of Acute Myeloid Leukemia (AML), Acute Lymphoblastic Leukemia (ALL), Chronic Lymphoblastic Leukemia (CLL), Mantle Cell Lymphoma (MCL), and Multiple Myeloma (MM) models
-
Well-characterized models featuring patient responses, clinical annotation, NGS data (WES and RNASeq), deep phenotyping using flow cytometry, proteomics, and phospho-proteomics datasets available in Lumin
Data-Driven Insights to Accelerate Your Hematologic Program
Hematological VitroScreen Endpoints








"Champions Oncology’s Hematological VitroScreen has become a critical tool in our research arsenal. The platform’s high throughput and precise results have significantly improved our ability to screen and analyze potential therapies for blood cancers."
___________________________________________________
"The Hematological VitroScreen from Champions Oncology has significantly enhanced our ability to evaluate drug efficacy and safety. The platform’s comprehensive data and high sensitivity have provided us with crucial insights into hematologic malignancies and drug interactions."
___________________________________________________
The Autologous AML Platform is an ex vivo co-culture assay developed and optimized to interrogate the responses of your immuno-oncology (IO) drugs in only 4 days using primary patient models.
In this study, we present an integrative analysis of multiple omic datasets coupled with phenotypic and therapeutic response profiles of Cytarabine from a cohort of primary Acute Myeloid Leukemia (AML) tumors, and Olaparib from a cohort of Patient-Derived Xenograft (PDX) models of ovarian cancer. These analyses, termed Pharmaco-Pheno- Multiomic (PPMO) Integration, established novel complex biomarker profiles that were used to accurately predict prospective therapeutic response profiles in cohorts of newly profiled AML and ovarian tumors.


