NSCLC TumorGraft3D Screen
Champions' new NSCLC TumorGraft3D Screen includes 22 NSCLC models, featuring EGFR-mutant, KRAS-mutant, EGFR+ MET+ models, and models with acquired resistance to osimertinib. These models are well-characterized with patient responses, clinical annotation, proliferation status, and SOC response.
Using Champions' innovative TumorGraft3D Platform, a matrix-free assay with proprietary media, this screen allows for the formation and maintenance of TumorGraft3D. It can rank the effects of different test agents across multiple models, investigate tumor cell biology and its interaction with the tumor microenvironment, and assess therapeutic agent synergy in experimental combinations.
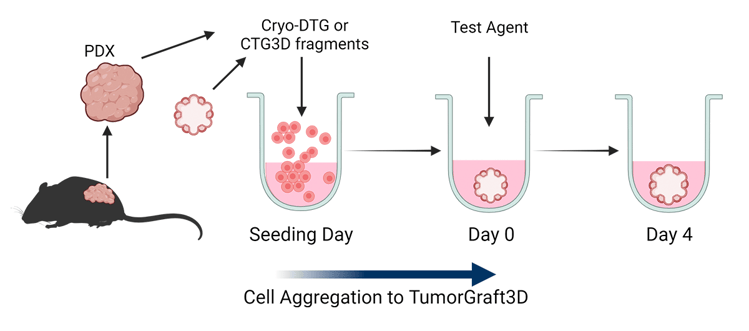
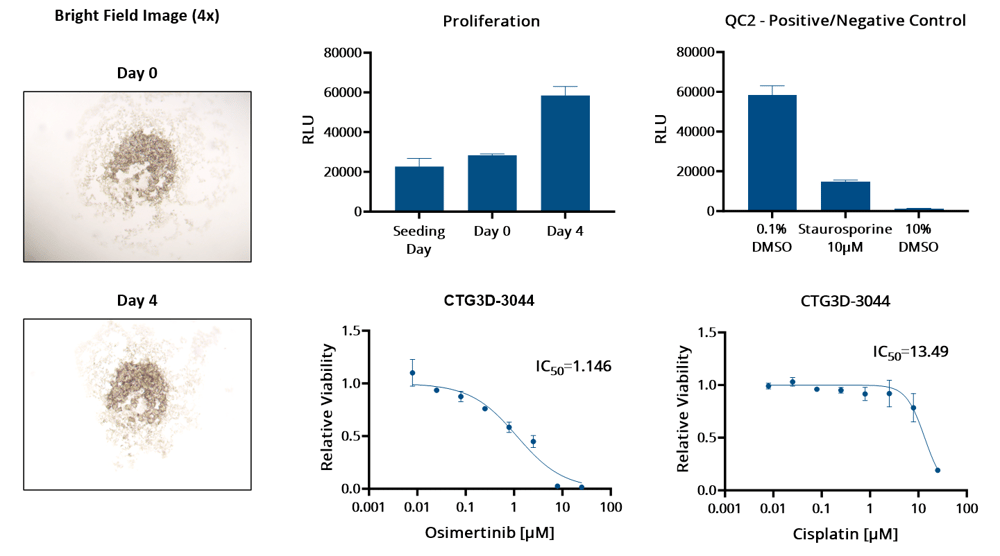
Characterization of NSCLC TumorGraft3D models CTG3D-3044: bright field images confirm 3D structure formation; proliferation demonstrate model viability for the duration of the assay and that this model proliferates during the dosing window*; positive/negative controls prove reliability of the assay; dose response curves show that the model is highly sensitive to osimertinib, but reveals a lower sensitivity to cisplatin.
*Models included in the screen are low passage TumorGraft3D derived from low passage PDX models and show a proliferation rate variability that well represents the variability observed in patients' tumors.
TumorGraft3D Screen Highlights:
- Screen provides additional cost-saving compared to a stand-alone study
- 22 NSCLC TumorGraft3D models
- no minimum to enroll
- 50% off of an SOC arm (selected by Champions)
- Included Endpoint: CellTiter-Glo® readout to evaluate cell viability/proliferation with an option to include high-content imaging analysis
- Luminex endpoint analysis can be evaluated at an additional cost
- Custom bioinformatics analyses, or NGS data licensing are available upon request.
